
Compliance Tool: Model Whistleblower Protection Policy
This model can help you create a whistleblower protection policy for your lab to encourage employees to voice their compliance concerns.

This model can help you create a whistleblower protection policy for your lab to encourage employees to voice their compliance concerns.

A False Claims Act whistleblower lawsuit can do significant damage to your lab and its reputation, even if the case lacks truth and merit.

Although the law officially took effect on March 29, the FDA says it isn’t planning to enforce the new rules until October 1.
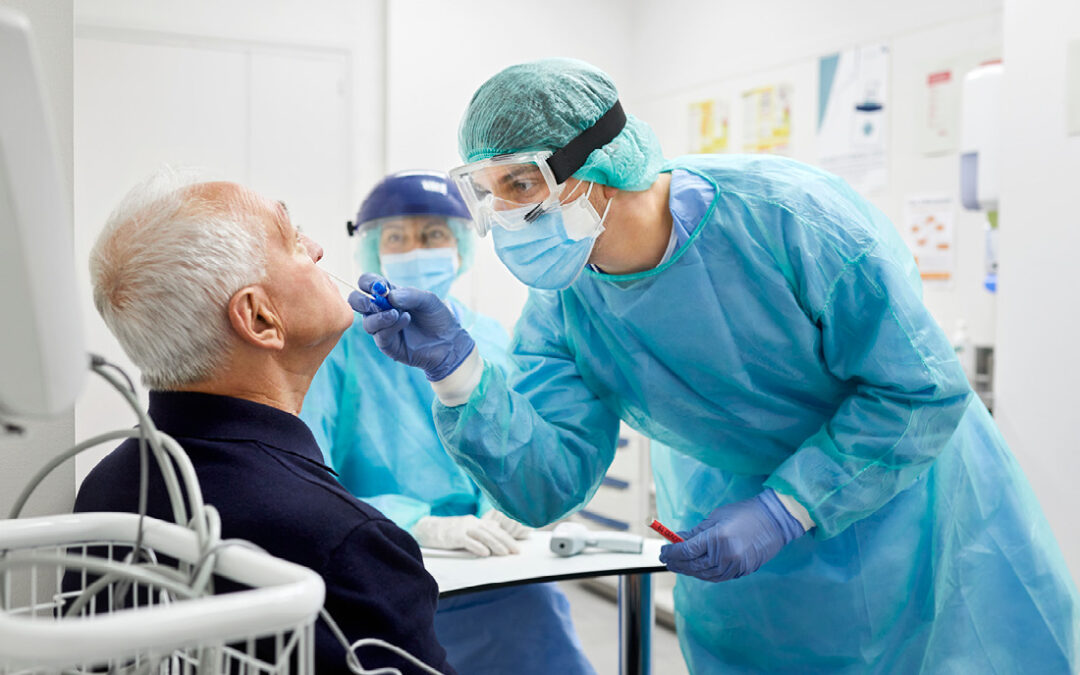
In the latest major COVID-19-related enforcement action, 18 individuals were charged in various schemes worth more than $490 million.

Recently released NGS systems will be helpful to researchers but are unlikely to immediately impact diagnostics, expert says.

COVID-19 test makers will either have to get full marketing approval, or take the product off the market.

Labs must use the new Advance Beneficiary Notice of Noncoverage (ABN) form starting on June 30, 2023, when the old version expires.
New document aims to help clinicians navigate screening guidelines, choose testing strategies, and analyze test results.
On March 27, the agency released a new guidance document to help test developers make the transition to full approval.

The inexpensive POC device is a biosensor that performs COMT genotyping as a biomarker for hypnotizability.