Keep Informed on Legal and Compliance Developments Affecting Diagnostic Labs
Best practices and expert insight to help lab professionals comply with regulations and identify strategic trends in business and technology
Already a Subscriber? Log in Here
Lab Industry Advisor Subscription
Struggling with Staying Informed? In the fast-paced world of diagnostic medicine, staying updated with the latest legal, compliance, regulatory, and industry insights is crucial. Yet, finding reliable and comprehensive information can be overwhelming and time-consuming.
Lab Industry Advisor to the Rescue! Lab Industry Advisor provides comprehensive, up-to-date insights and analyses tailored to your needs. With expert commentary and actionable advice, you’ll stay ahead of the curve. No more sifting through endless sources—everything you need is right here!
Start your free trial today and see how Lab Industry Advisor can transform your approach. Get instant access to invaluable resources designed to keep you informed and competitive. Explore detailed reports, in-depth articles, and exclusive industry insights that help you make better decisions faster. Choose from three subscription levels tailored to fit your needs: Essential, Premium, and Elite.






Additional Business Intelligence Offerings
Lab Industry Advisor Subscriptions Include

Analysis and insight on key compliance, legal, and business developments affecting laboratories
- Updates on changes to regulations
- Healthcare-related laws
- FDA approvals
- Business deals in the diagnostics space, and more
Latest Articles
New FDA LDTs Guidance Offers Useful Summary, But Questions Remain
Recently released document for small labs offers a more digestible version of the final rule requirements but still lacks key details
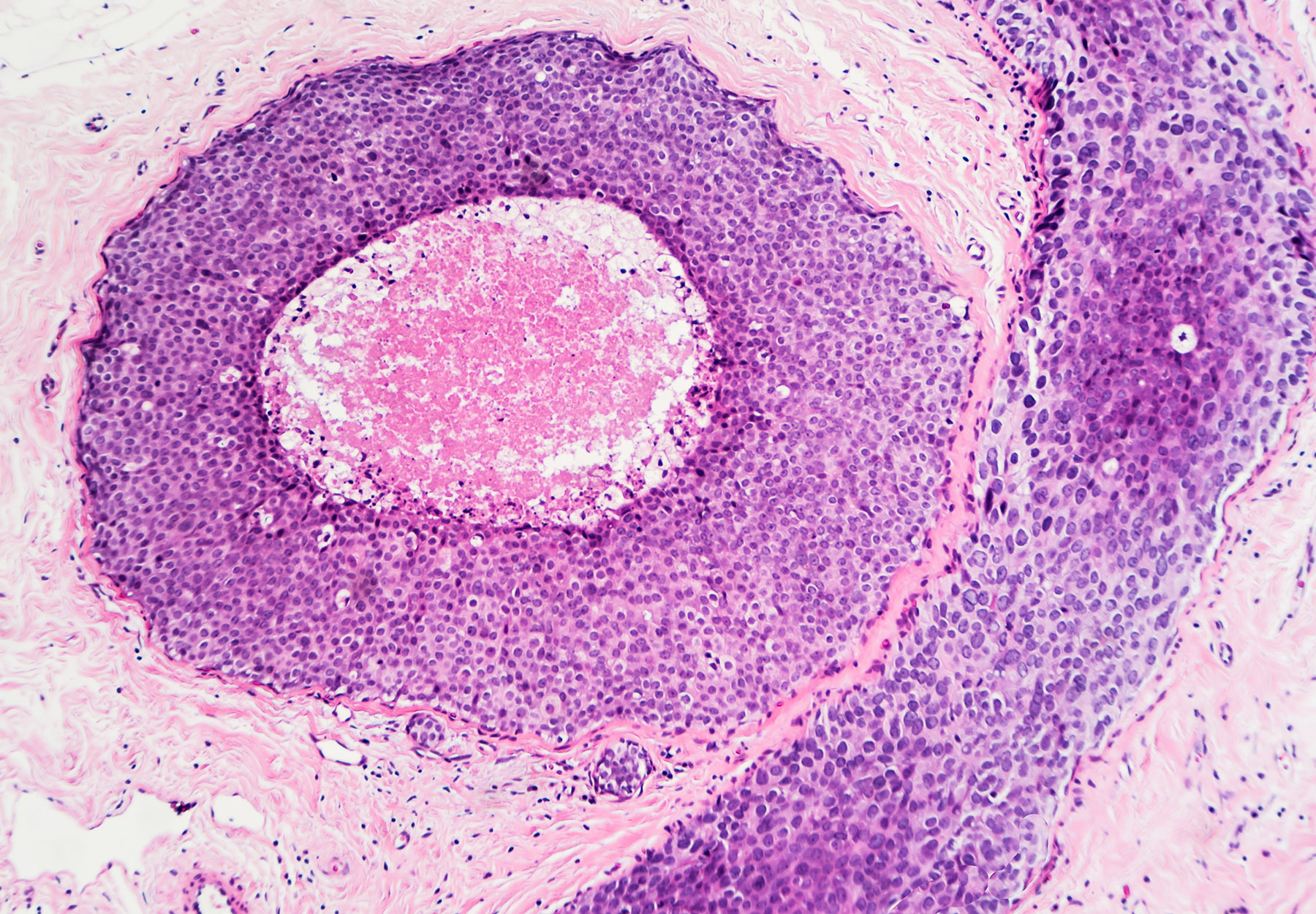
Deep Learning Poised to Improve Breast Cancer Imaging
WASHINGTON, DC — Researchers have developed a new image reconstruction approach that could contribute to better breast cancer detection. The deep learning algorithm overcomes a major hurdle in multi-modality imaging by allowing images to be recovered in real time....
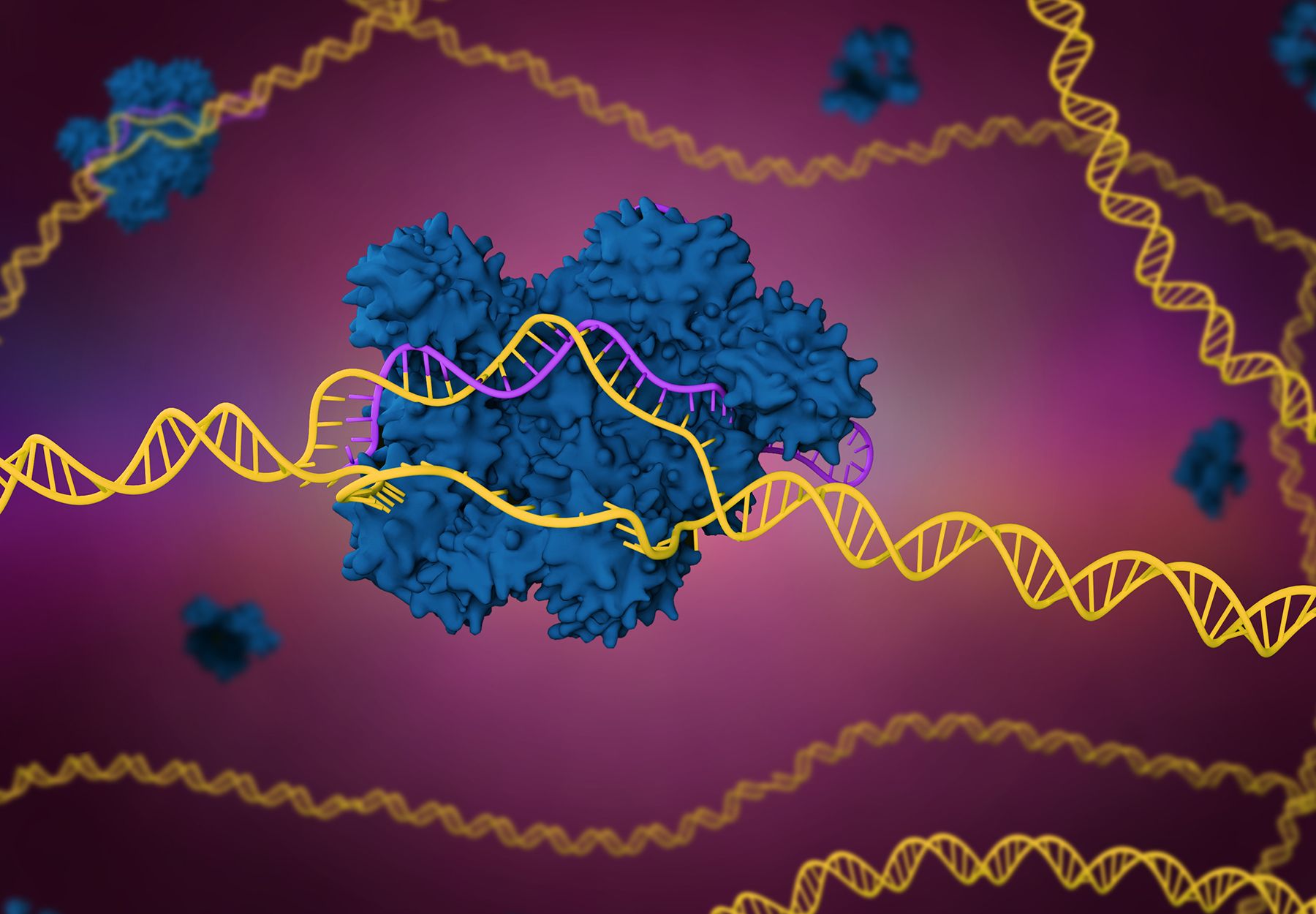
Element and Jumpcode Team Up to Boost Many Genomic Applications
SAN DIEGO, CA — Element Biosciences, Inc., developer of a new and disruptive DNA sequencing platform, today announced a collaboration and co-marketing partnership with Jumpcode Genomics that validates Jumpcode’s CRISPRclean technology on Element’s AVITI System. The...
Feds Target Fraudulent Billing of Genetic Cancer Tests
False ordering and billing of cancer genetic (CGx) tests has become a favorite target for federal fraud enforcers, with the U.S. Department of Justice (DOJ) reporting two notable convictions in the closing months of 2021. On Nov. 17, a 57-year-old former lab sales...
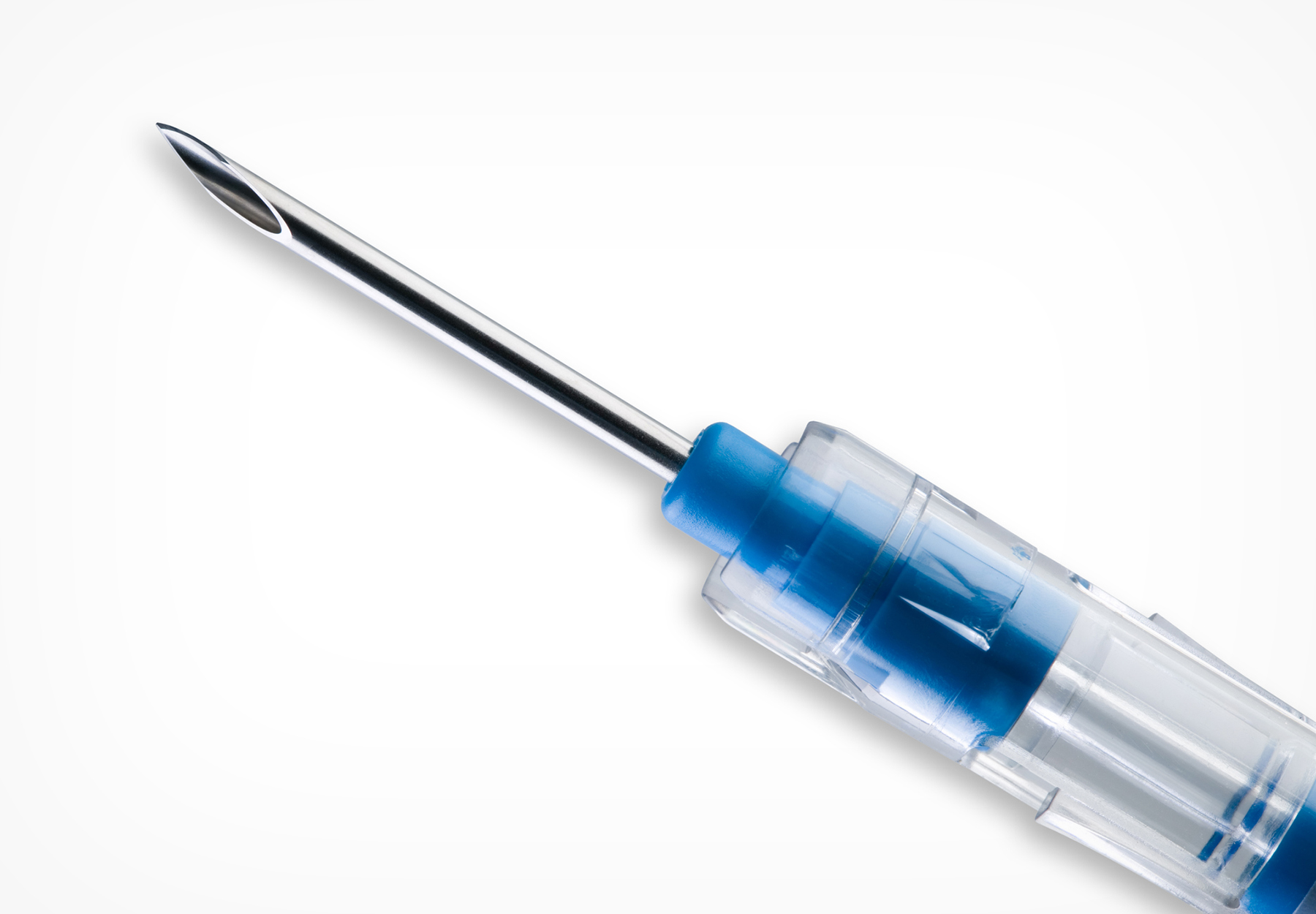
Healthy Growth Predicted for Global Aspiration & Biopsy Needles Market
According to a recently released report from market research company Research and Markets, the global aspiration and biopsy needles market is poised for solid growth over the next five years. The report valued the market at an estimated $879 million US for 2021 and...
Labcorp Completes Acquisition of Personal Genome Diagnostics
BURLINGTON, NC — Labcorp, a leading global life sciences company, announced Feb. 21 that it has closed its acquisition of Personal Genome Diagnostics Inc. (PGDx), a leader in cancer genomics with a portfolio of comprehensive liquid biopsy and tissue-based products....
Startup Awarded NIH Grant to Develop Endometriosis Test
OAKLAND, CA — NextGen Jane, a genomics startup generating big data for novel applications in molecular diagnostics and drug development for female reproductive disorders, announced that it has received the green light for the second phase of a Small Business...
2021 Was a Breakthrough Year for Breakthrough Devices Program
The FDA designated 213 submissions as breakthrough devices in 2021, the most awarded in a single year, by far.
Beyond EUA: FDA Grants 510(k) Clearance for a COVID-19 Test
On Jan. 27, BioFire Defense's COVID-19 Test 2, an assay developed in collaboration with the U.S. Army, received 510(k) clearance from the FDA. Previously, the test had been cleared only for emergency use. And that’s a big deal. In fact, almost all of the more than...