Keep Informed on Legal and Compliance Developments Affecting Diagnostic Labs
Best practices and expert insight to help lab professionals comply with regulations and identify strategic trends in business and technology
Already a Subscriber? Log in Here
Lab Industry Advisor Subscription
Struggling with Staying Informed? In the fast-paced world of diagnostic medicine, staying updated with the latest legal, compliance, regulatory, and industry insights is crucial. Yet, finding reliable and comprehensive information can be overwhelming and time-consuming.
Lab Industry Advisor to the Rescue! Lab Industry Advisor provides comprehensive, up-to-date insights and analyses tailored to your needs. With expert commentary and actionable advice, you’ll stay ahead of the curve. No more sifting through endless sources—everything you need is right here!
Start your free trial today and see how Lab Industry Advisor can transform your approach. Get instant access to invaluable resources designed to keep you informed and competitive. Explore detailed reports, in-depth articles, and exclusive industry insights that help you make better decisions faster. Choose from three subscription levels tailored to fit your needs: Essential, Premium, and Elite.






Additional Business Intelligence Offerings
Lab Industry Advisor Subscriptions Include

Analysis and insight on key compliance, legal, and business developments affecting laboratories
- Updates on changes to regulations
- Healthcare-related laws
- FDA approvals
- Business deals in the diagnostics space, and more
Latest Articles
LDTs Rule Update: More Support for Legal Challenges Against FDA
Key clinical laboratory organizations file amicus brief, reiterating that the FDA is overstepping its authority, and its new rule would cause “irreparable patient harm”
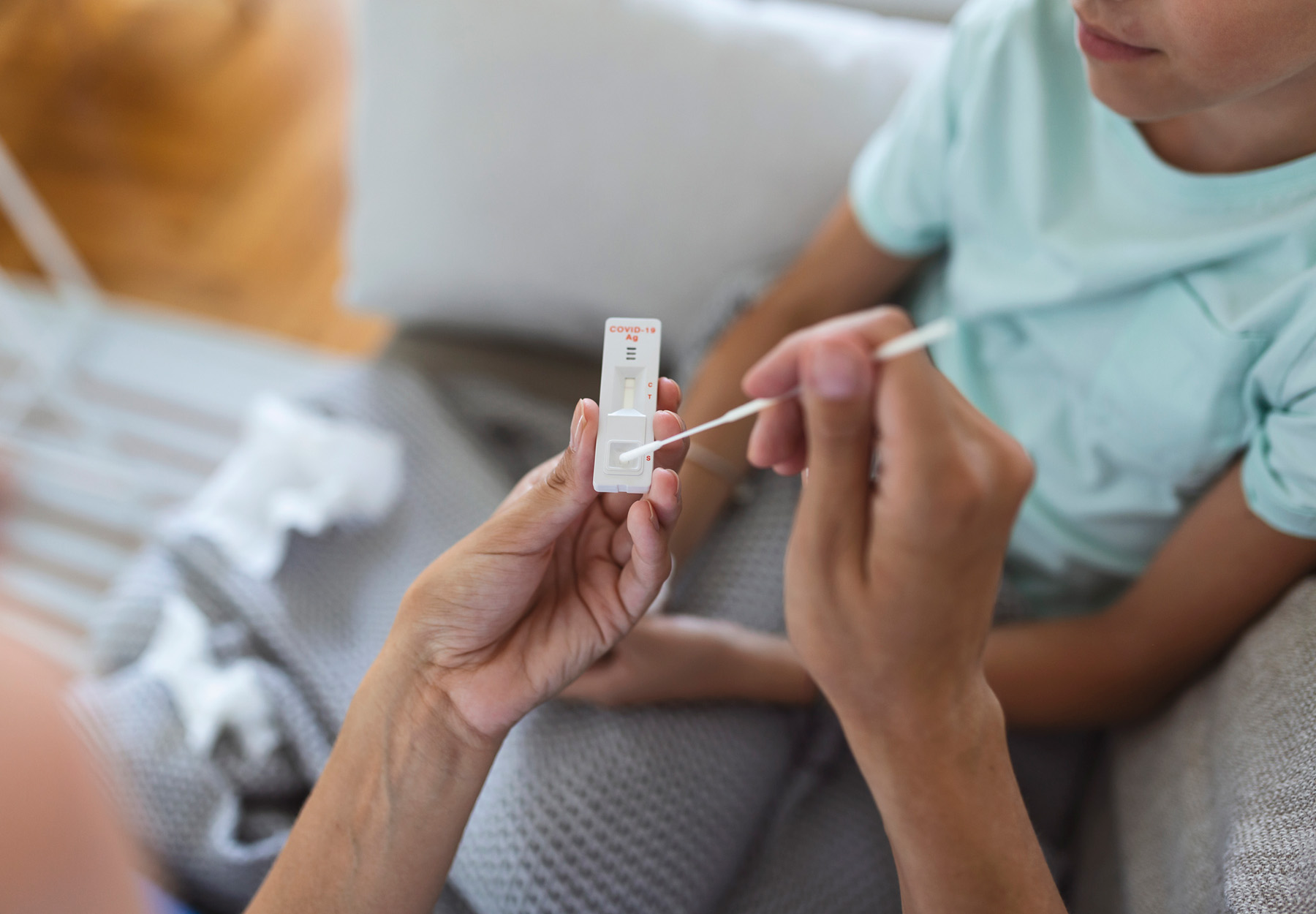
FDA Extends Shelf Life of At-Home Rapid COVID-19 Tests
Users should realize that the FDA can extend the shelf life of a test when and if it receives additional data supporting such an extension.
FY 2022 Q2 Earnings Trend Down, but Much Less than Predicted
While not nearly as robust as a year ago, diagnostic companies’ revenues held up surprisingly well in Q2 2022.
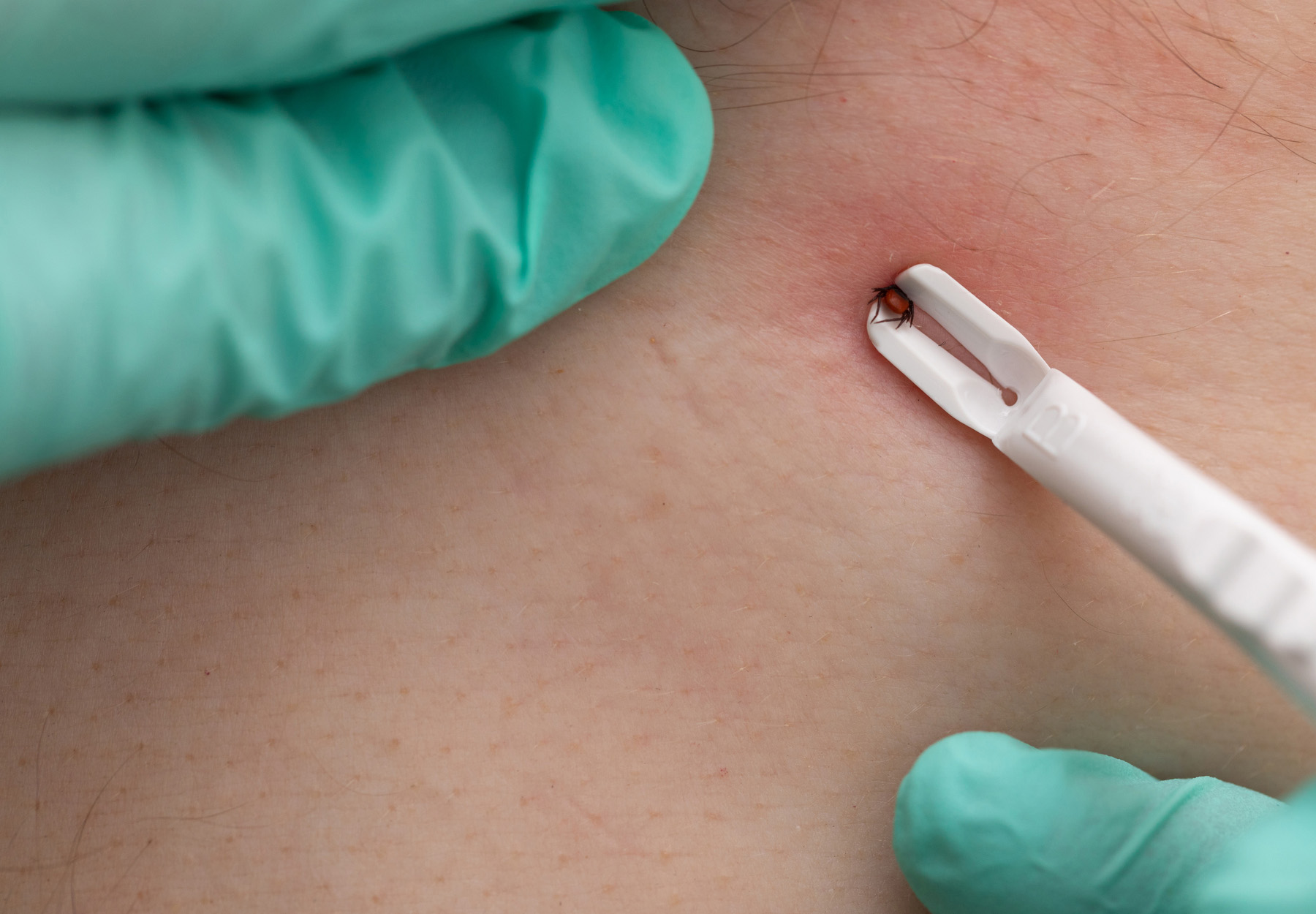
Is an Antibody Test for Early Detection of Lyme Disease Feasible?
Recent study suggests a new test may help doctors detect Lyme disease during the early stages when treatment is likely to be more effective.
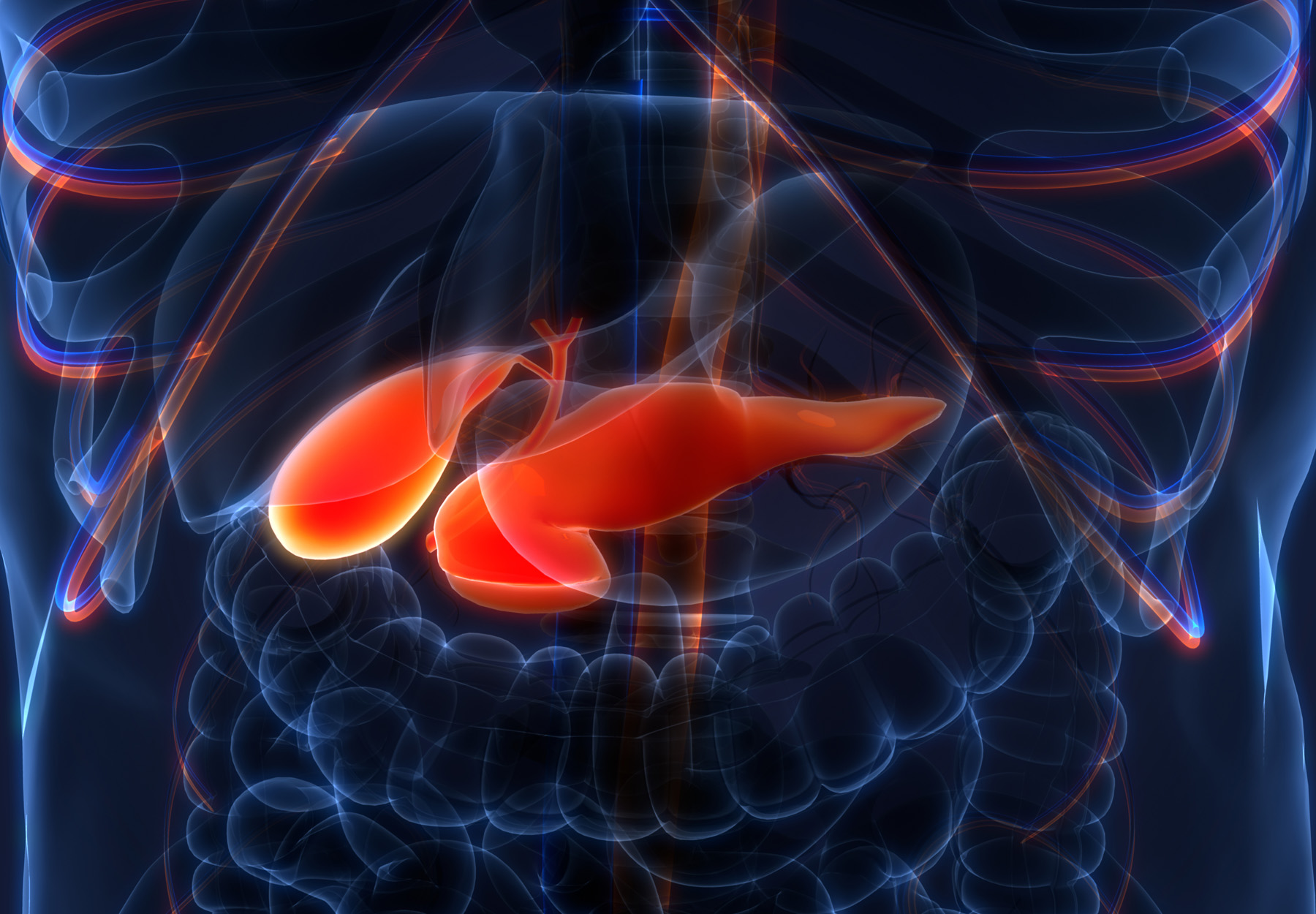
Pancreatic Cancer Is Caught Sooner When Regular Screening Done
A recent study shows that most patients who underwent such screening were diagnosed at an earlier stage than those who did not.
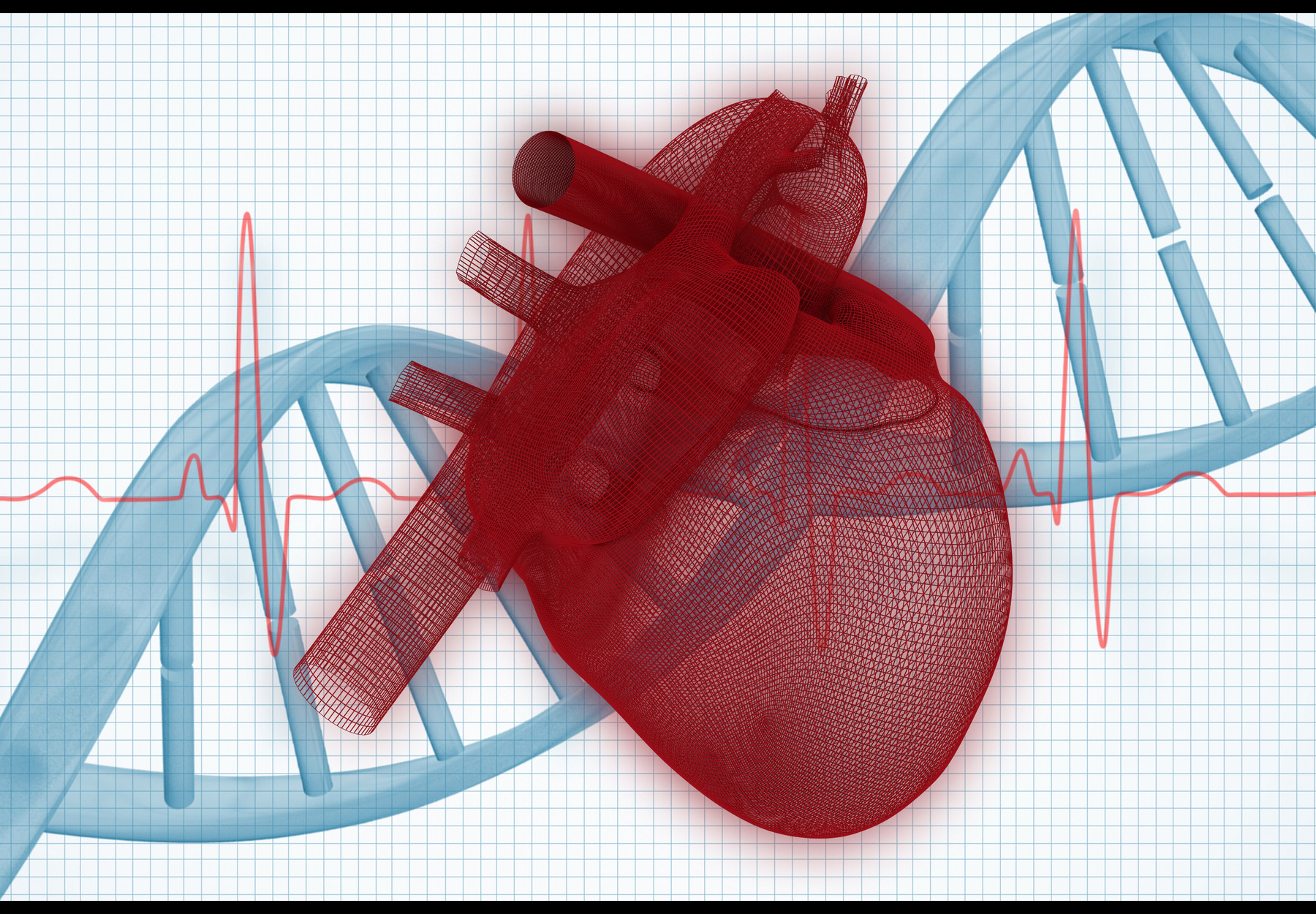
Combining Certain Genetic Tests for Heart Conditions Provides Better Results
A recent study shows that combined cardiomyopathy and arrhythmia genetic testing catches things disease-specific testing would miss.
New Guidelines Recommend Molecular Testing for Pediatric Brain Cancer Treatment
For labs, the big takeaway is how prominently molecular testing and profiling figures in so many aspects of the NCCN’s recommendations.
Repeat At-Home COVID-19 Antigen Tests to Avoid False Negatives, FDA Says
The agency is advising people to perform repeat, or serial testing after a negative result, regardless of symptoms.
Telehealth Doesn’t Discourage In-Person Care or Drive Wasteful Use
Two recent studies show that most people see telehealth as a supplement to in-person visits, rather than a replacement.
Getting Paid: How to Complete the ABN Form
Completing the Advance Beneficiary Notice of Noncoverage form can be extremely complex; this guide helps simplify the process.
Inform Diagnostics Pays $16M to Settle False Billing Allegations
In this Labs in Court monthly roundup, the Texas-based pathology lab was accused of falsely billing Medicare for unnecessary tests on biopsy specimens.