Keep Informed on Legal and Compliance Developments Affecting Diagnostic Labs
Best practices and expert insight to help lab professionals comply with regulations and identify strategic trends in business and technology
Already a Subscriber? Log in Here
Lab Industry Advisor Subscription
Keeps you up to date on the latest legal, compliance, and business developments affecting the lab industry, while offering analysis and insight to ensure your lab’s success.






Additional Business Intelligence Offerings
Lab Industry Advisor Subscriptions Include

Analysis and insight on key compliance, legal, and business developments affecting laboratories
- Updates on changes to regulations
- Healthcare-related laws
- FDA approvals
- Business deals in the diagnostics space, and more
Latest Articles
New FDA LDTs Guidance Offers Useful Summary, But Questions Remain
Recently released document for small labs offers a more digestible version of the final rule requirements but still lacks key details
Israeli Startup Salignostics Launches First Saliva-Based Pregnancy Test
The company has successfully completed clinical trials in Israel on more than 300 women and is applying for CE marking and FDA approval.
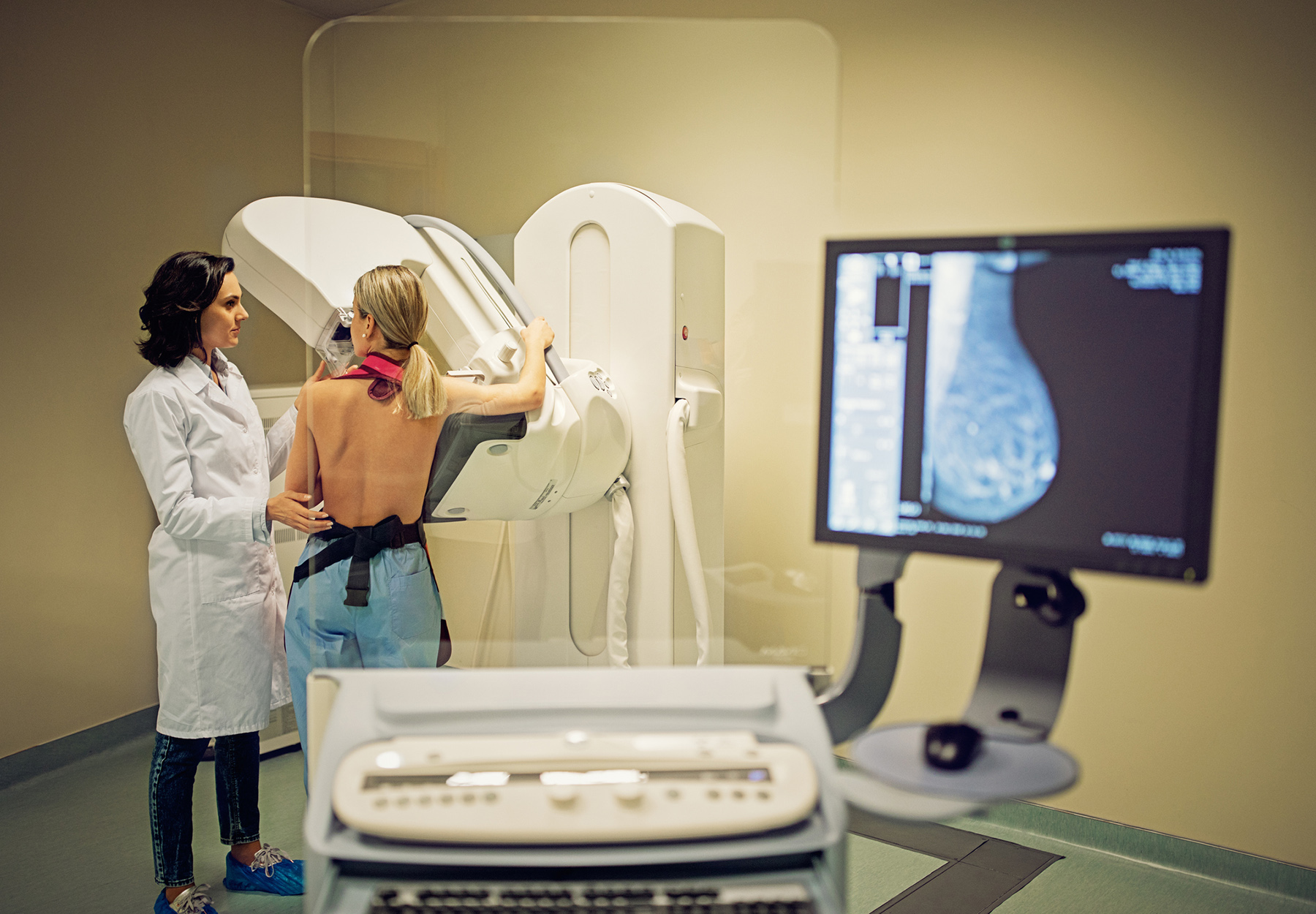
Minorities See Biggest Drops in Cancer Screening During the Pandemic
New research shows cancer screening prevalence declined in 2020 compared to 2018, though not for all cancer types studied.
FDA Warns of Cybersecurity Vulnerability in Certain NGS Instruments
A glitch in the local run management software may open the door to hacking, agency says.
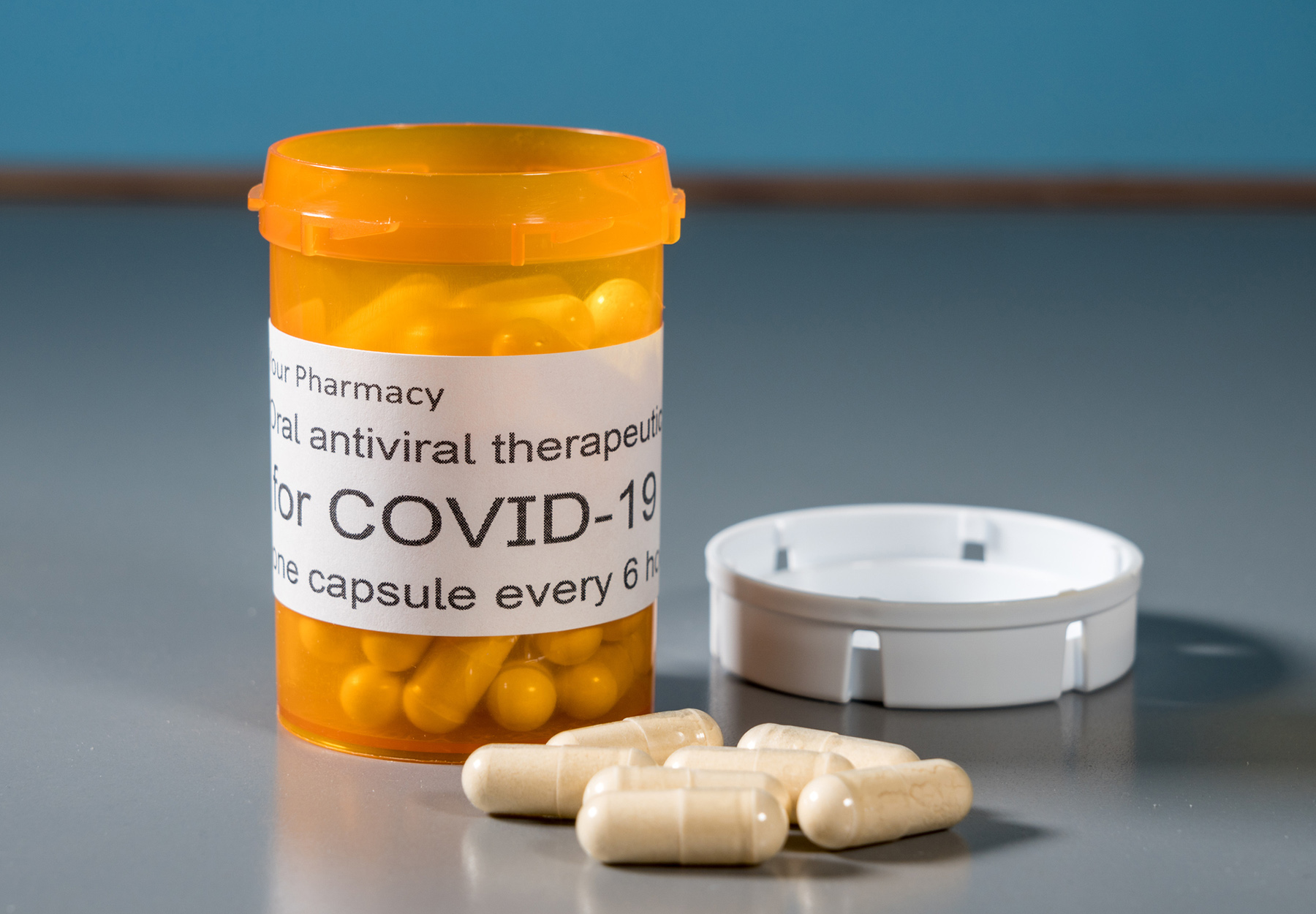
Test-To-Treat Locations Now Up to 2,500
White House releases an update on the progress of the initiative to provide equitable access to COVID-19 testing and treatments in the US.
Twenty Percent of DME Suppliers Non-Compliant When Billing Medicare
OIG report finds that $8 million of $40 million in claims made by such suppliers in a recent audit did not meet Medicare requirements.
Does Offering Free Access to Drug Pricing Tool Cross the Kickback Line?
NY federal court ruling sheds light on what constitutes “remuneration” banned by the Anti-Kickback Statute.
Illumina and Precision Health Team Up for Population Study
Partnership will make use of Illumina’s capabilities and expertise in large-scale genomic sequencing and quickly generate high-quality data.
New Senate Bill Targets PBM Consumer Abuses
Lawmakers blame pharmacy benefit managers for high drug prices, introducing a bill to prevent what they term unfair practices.
The Sun Sets on the Controversial SUNSET Rule
HHS pulls the plug on a Trump rule requiring regulations to expire automatically after 10 years.
DOJ Adds 6 Texas Physicians to Improper Lab Test Claims Complaint
In this week’s Labs in Court roundup, six physicians were accused of billing for unnecessary lab tests in connection with an earlier lab fraud complaint.