Keep Informed on Legal and Compliance Developments Affecting Diagnostic Labs
Best practices and expert insight to help lab professionals comply with regulations and identify strategic trends in business and technology
Already a Subscriber? Log in Here
Lab Industry Advisor Subscription
Keeps you up to date on the latest legal, compliance, and business developments affecting the lab industry, while offering analysis and insight to ensure your lab’s success.






Additional Business Intelligence Offerings
Lab Industry Advisor Subscriptions Include

Analysis and insight on key compliance, legal, and business developments affecting laboratories
- Updates on changes to regulations
- Healthcare-related laws
- FDA approvals
- Business deals in the diagnostics space, and more
Latest Articles
New FDA LDTs Guidance Offers Useful Summary, But Questions Remain
Recently released document for small labs offers a more digestible version of the final rule requirements but still lacks key details
FDA Orders LuSys and Celltrion to Recall Unauthorized COVID-19 Rapid Tests
This week, two companies announced Class I recalls, the most serious kind, for COVID-19 antigen and antibody tests that have reached the US market without FDA Emergency Use Authorization (EUA) or 510(k) clearance. San Diego-based LuSys Laboratories is recalling...
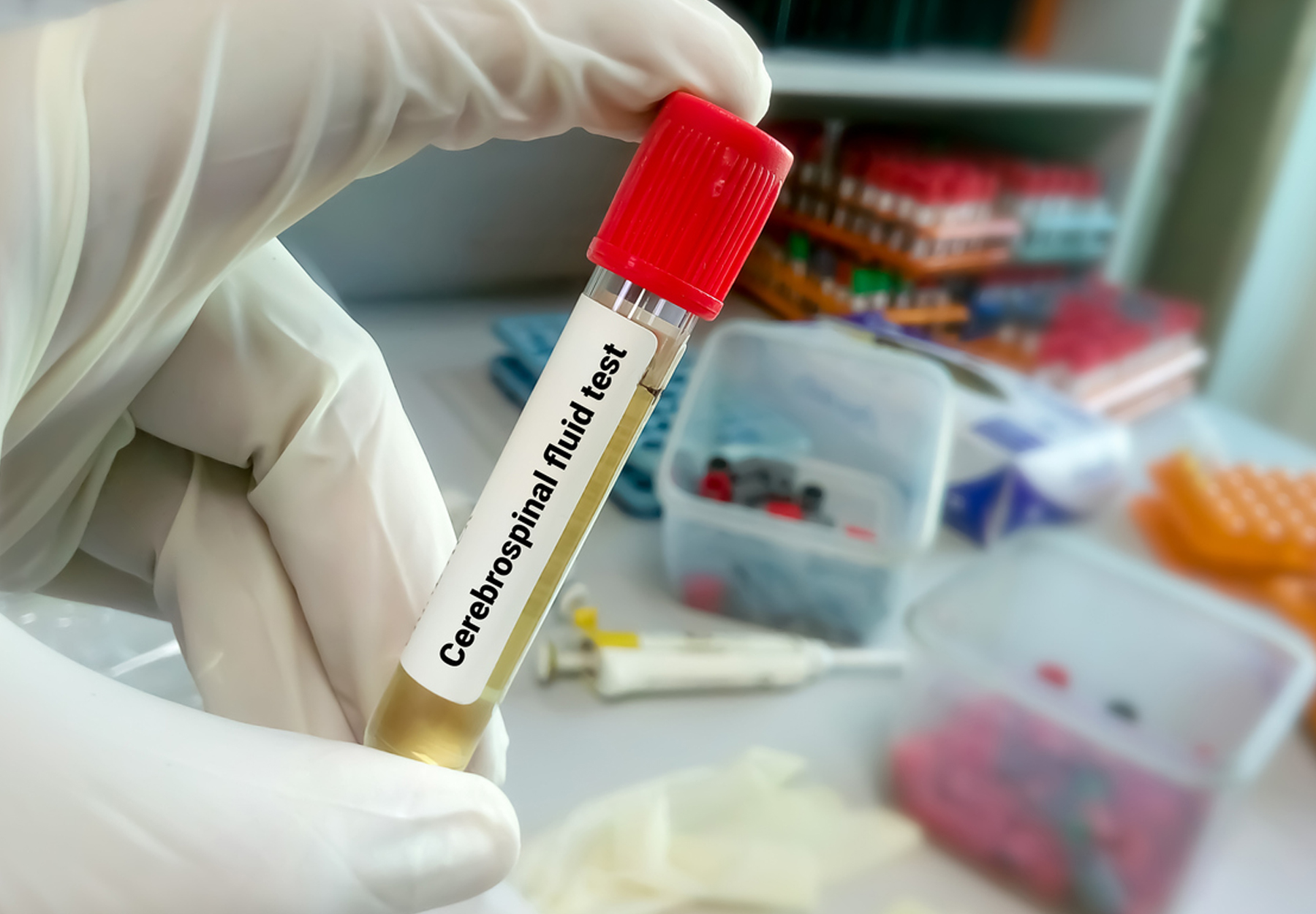
How CSF Tests May Help Recruitment for Alzheimer’s Drug Clinical Trials
A recent study shows these tests may make it easier and more cost-effective to identify patients for studies of new PET treatments.
After Steady Decline, Telehealth Use Surges during Omicron
When the pandemic and lockouts first began, telehealth utilization soared to unprecedented levels. But while telehealth use remains well above pre-pandemic levels, thanks in part to relaxed Medicare coverage rules, the initial wave steadily ebbed starting in May...
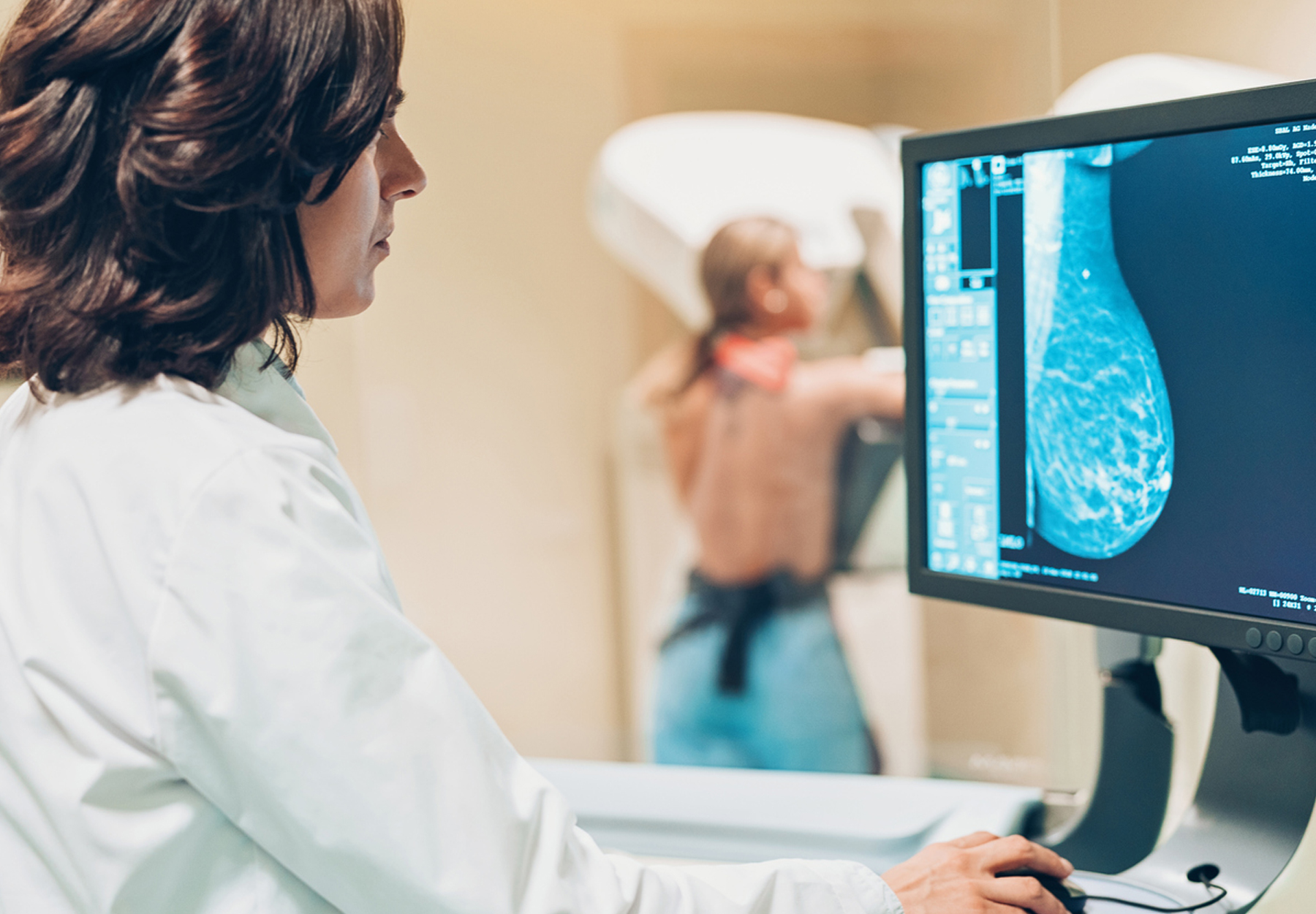
FDA Grants Approval to Breast Cancer Treatment and Companion Diagnostic
Certain cancer patients now have access to a new treatment option and companion diagnostic. On March 11, the U.S. Food and Drug Administration approved olaparib (Lynparza, AstraZeneca Pharmaceuticals, LP) for treating adults with deleterious or suspected...
US Government Launches Nationwide Test-to-Treat Initiative
Recently unveiled program aims to ensure easy access to COVID-19 treatments for all eligible Americans.
Physicians Get a Break on 2021 MIPS Payment Adjustments
Just as it seemed like physician practice patterns were getting back to normal, the late-year Omicron surge skewed the Merit-based Incentive Payment System (MIPS) quality data for 2021. With that in mind, the Centers for Medicare & Medicaid Services (CMS) has...
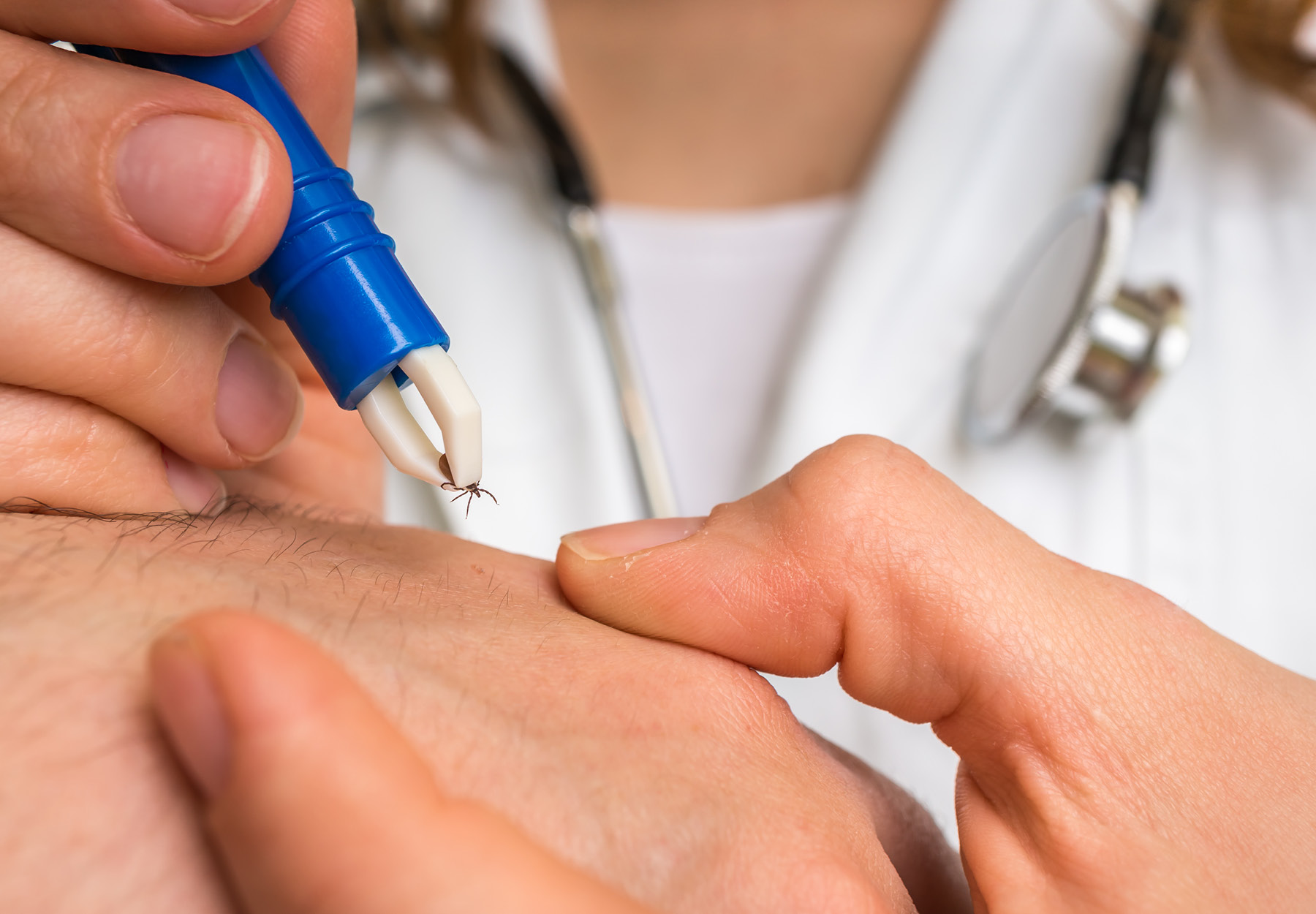
Potential New Test Could Diagnose Lyme Disease Sooner
For scientists and clinicians alike, one of the Holy Grails for successfully treating and curing Lyme disease is developing tests that identify the disease sooner, show when people are cured of infection, and can diagnose reinfection. Now, researchers at Tufts...
FDA and Industry Reach Agreement on MDUFA V Medical Device User Fees
For a while, it looked like the FDA and medical tech industry might not cut a deal on the next round of medical device user fees under the Medical Device User Fee Amendments (MDUFA V) before the current agreement expires in September. But last week, the agency...
Singleron Extends Reach outside of China with Acquisition of Proteona
Last week, Chinese genomics company Singleron Biotechnologies, which develops single-cell multi-omics platform technologies and helps translate them to the clinic, announced it had acquired Proteona Pte. Ltd. Based in Singapore, Proteona is also focused on the...