LDT Oversight Remains Under CLIA, for Now
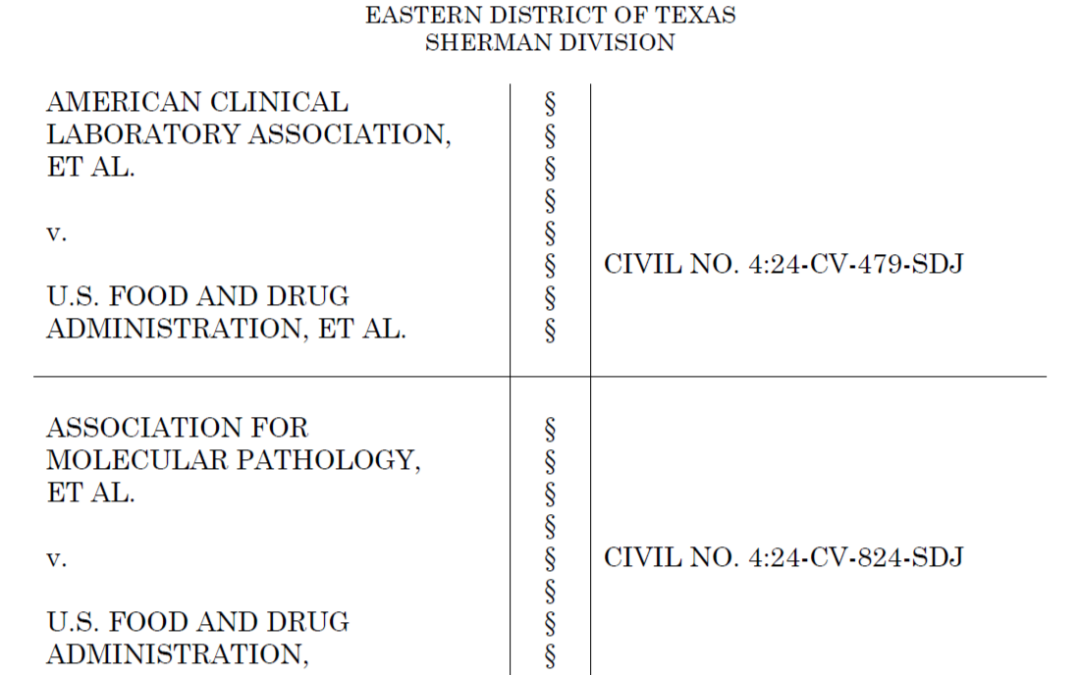
Judge rules that the FDA has ‘no authority’ to issue its rule governing laboratory-developed tests

Judge rules that the FDA has ‘no authority’ to issue its rule governing laboratory-developed tests

Put into place late last year, the new rules place more emphasis on formal and continuing education requirements

The 2025 CLFS update—what has changed, what has stayed the same, and what that means for diagnostic test reimbursement in 2025

Experts say new leadership could impact the cost and availability of IVDs as well as patients’ ability to afford lab testing

How labs form an indispensable part of the CMS National Quality Strategy, helping to drive healthcare improvements and scientific growth.